CRISPR Therapies Move Into Larger Multi-Center Trials
Several CRISPR-based treatments for blood disorders and rare genetic diseases have entered broader multi-center evaluations this month, marking a cautious but meaningful expansion in gene-editing research. These trials build on earlier safety studies that demonstrated stable editing in targeted cells, though long-term impacts are still being closely monitored.
Stylized CRISPR-Cas9 illustration showing DNA strands being cut. Image credit: Ernesto del Aguila III, National Human Genome Research Institute, NIH (public domain).
What the New Trials Are Designed to Measure
The expanded studies focus on conditions such as sickle cell disease, beta-thalassemia, and select immune disorders where a single genetic change plays a major role in symptoms. According to publicly available trial protocols, researchers are emphasizing three areas:
- Editing efficiency: How consistently the CRISPR system edits the intended gene across large patient samples.
- Durability: Whether edited cells continue functioning months to years after treatment.
- Safety markers: Monitoring for off-target edits, immune responses, or unexpected cellular changes.
The trials use standard clinical endpoints, including hemoglobin levels, transfusion independence, and validated quality-of-life measures. Researchers also collect extensive genomic data to track whether the edits remain stable over time.
Cautious Progress After Early-Stage Results
Earlier phase studies of several CRISPR therapies showed encouraging signs, particularly in blood disorders where edited stem cells produced sustained increases in functional blood components. However, investigators noted that most initial cohorts were small. As the trials widen to include multiple centers, the field will gain the larger datasets needed to evaluate consistency across different clinical environments.
Even so, researchers emphasize the importance of tempering expectations. While many patients in prior studies saw measurable improvement, sample sizes were limited, follow-up duration varied, and some treatment-related effects, such as temporary inflammation, require continued observation.
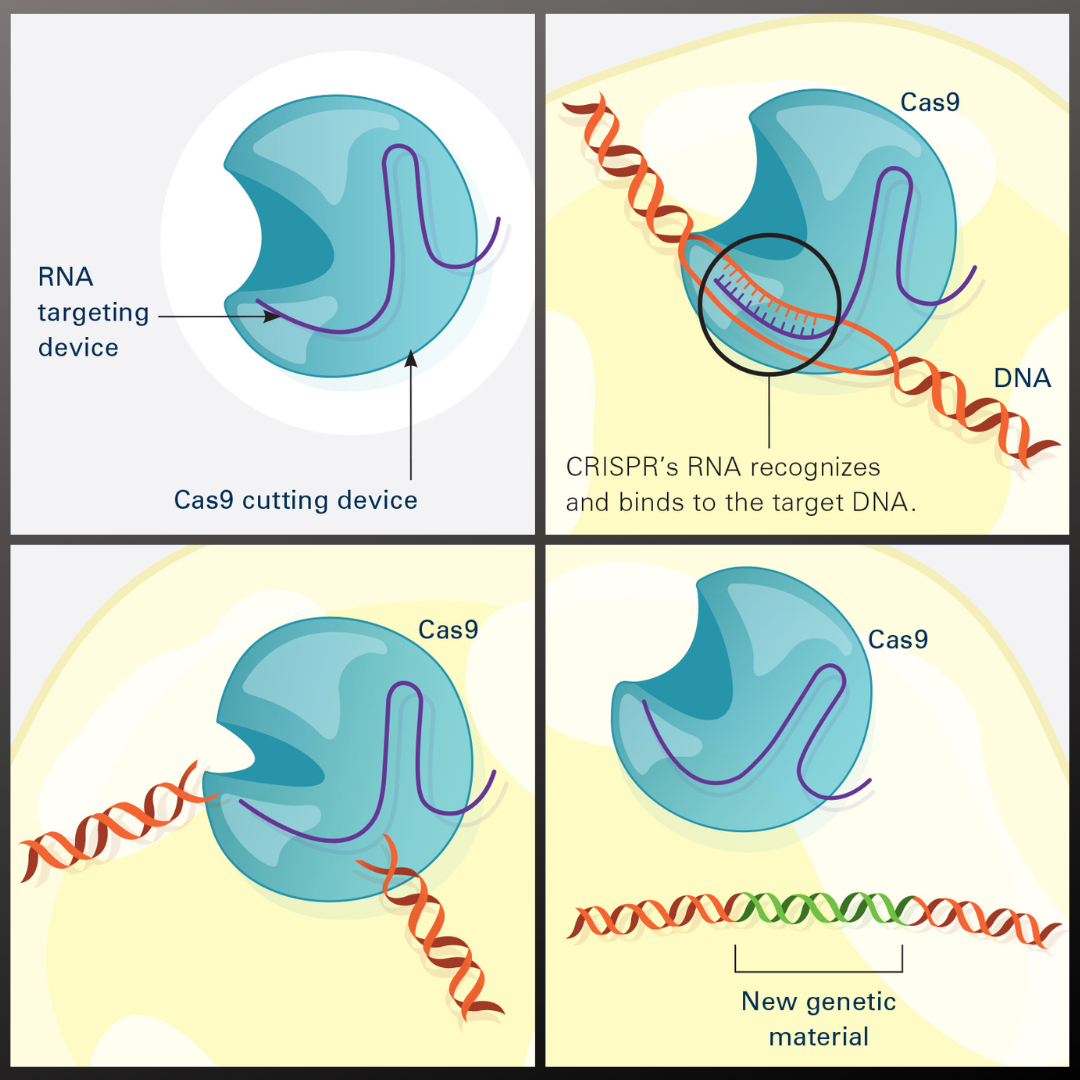
Illustration showing how the CRISPR-Cas9 system can be used as a gene-editing tool. Image credit: National Institute of General Medical Sciences, NIH (public domain).
Why These Trials Matter
The move toward multi-center evaluations signals growing confidence in both the underlying technology and the infrastructure required to deliver it. For conditions driven by well-characterized genetic mechanisms, CRISPR remains one of the most promising avenues for long-term therapeutic improvement. Still, investigators stress that these studies will determine whether early gains translate to broader populations.
Regulators have also highlighted the need for consistent reporting standards. Because CRISPR therapies involve altering cells before reinfusion, understanding the manufacturing and delivery process is as important as the genetic edit itself.
Remaining Unknowns
Despite the progress, significant questions remain. Long-term off-target effects are still being evaluated, and the durability of edits in certain immune or stem cell populations is not fully understood. Researchers also note that accessibility and cost will become important considerations if any therapy advances toward regulatory review.
As more data becomes available in 2026, the field will have a clearer picture of what CRISPR can reliably achieve in clinical practice. For now, the expansion of these trials represents a measured step forward, one grounded in observed results but still mindful of the limits of existing evidence.



Comments
Post a Comment